Medical Devices & Simulation
Regulatory-ready systems.
Aligned with: MedTech highlight
MedTech flagship system
Audit-ready ECMO training platform delivered in three modules: Housing, Vessel Integration, and the Final Combined Set.
Housing, Vessel Integration, and the Final Combined Set lock together for a clinically credible, production-ready training system.
Three-part product map
Final Combined Set = flagshipAutomation & validation support
AI-enabled manufacturing workflows, validation evidence, and audit-ready documentation.
Consultancy: EUR 499 / month · Product set: EUR 999 one-time

Flagship combined set
Complete ECMO training system
Regulatory alignment
Documentation flow and cleanroom discipline aligned to ISO expectations.
Advanced manufacturing
Medical-grade materials with automated quality control.
Service lines
Each lane supports the multidisciplinary story across MedTech, automation, and industrial delivery — aligned to the flagship and impact sections.
Connect the journey
Regulatory-ready systems.
Aligned with: MedTech highlight
Audit-ready workflows.
Aligned with: Business impact
Launch-ready media.
Aligned with: Portfolio showcase
Advisory & training.
Aligned with: FAQ + delivery flow
Certified cultivation.
Aligned with: Portfolio showcase
Pharma-grade supply.
Aligned with: Portfolio showcase
Controlled growth.
Aligned with: Portfolio showcase
Audit readiness.
Aligned with: Business impact
Performance control.
Aligned with: Portfolio showcase
Request a consolidated plan across technology, compliance, and launch.
Continuation of Medtech highlight
Modular ECMO training system with realistic flow layers, AI‑assisted validation, and compliance‑ready documentation.
Modular units
8+
Expandable core system.
Validation
Audit‑ready
Compliance documentation included.
Designed for hospitals and training centers, this all‑in‑one system delivers rapid setup, measured performance, and validated learning workflows.
What’s included
Core kitPrecision housing
Engineered shell and mounting.
Vascular layers
Bio‑mimic vessels and tissue.
AI oversight
Audit logs and QA analytics.
Implementation note
SupportCompatible with internal simulation labs and partner training programs. Deployment support available.
Language readiness
SupportEnglish, Türkçe, العربية available on request.
Atlas Global Technologies · Flagship pricing
A premium training platform with lifelike vascular integration, reusable components, and an AI admin panel. Multilingual support is available on request.
Aligned with MedTech program delivery & validation readiness
Flagship module visuals

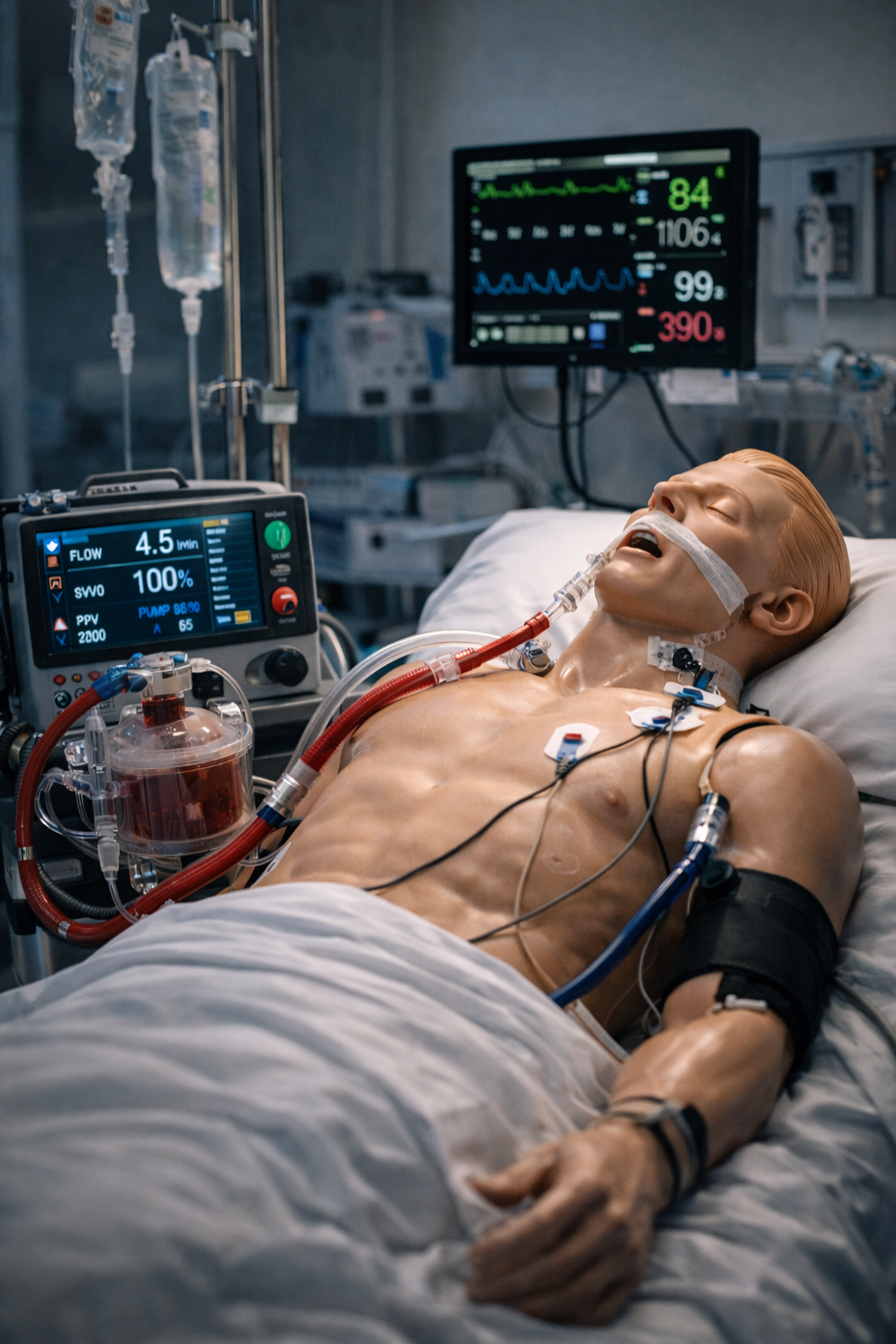
Housing product visual
Humanlike responsive housing shell
Clean anatomical form with a training‑grade finish.
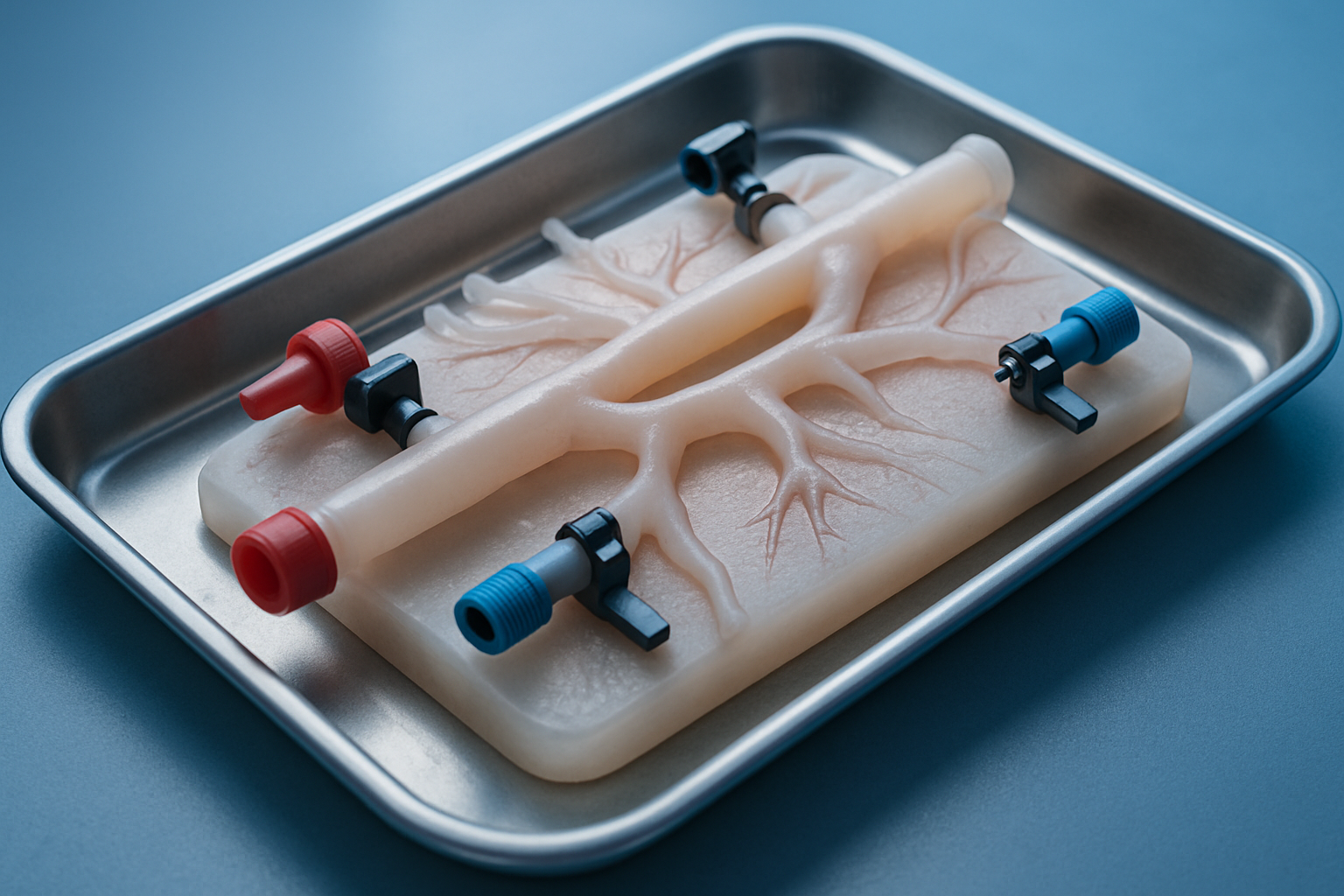
Vessel integration visual
Modular vascular circuit
Precision biotech detailing with realistic flow paths.
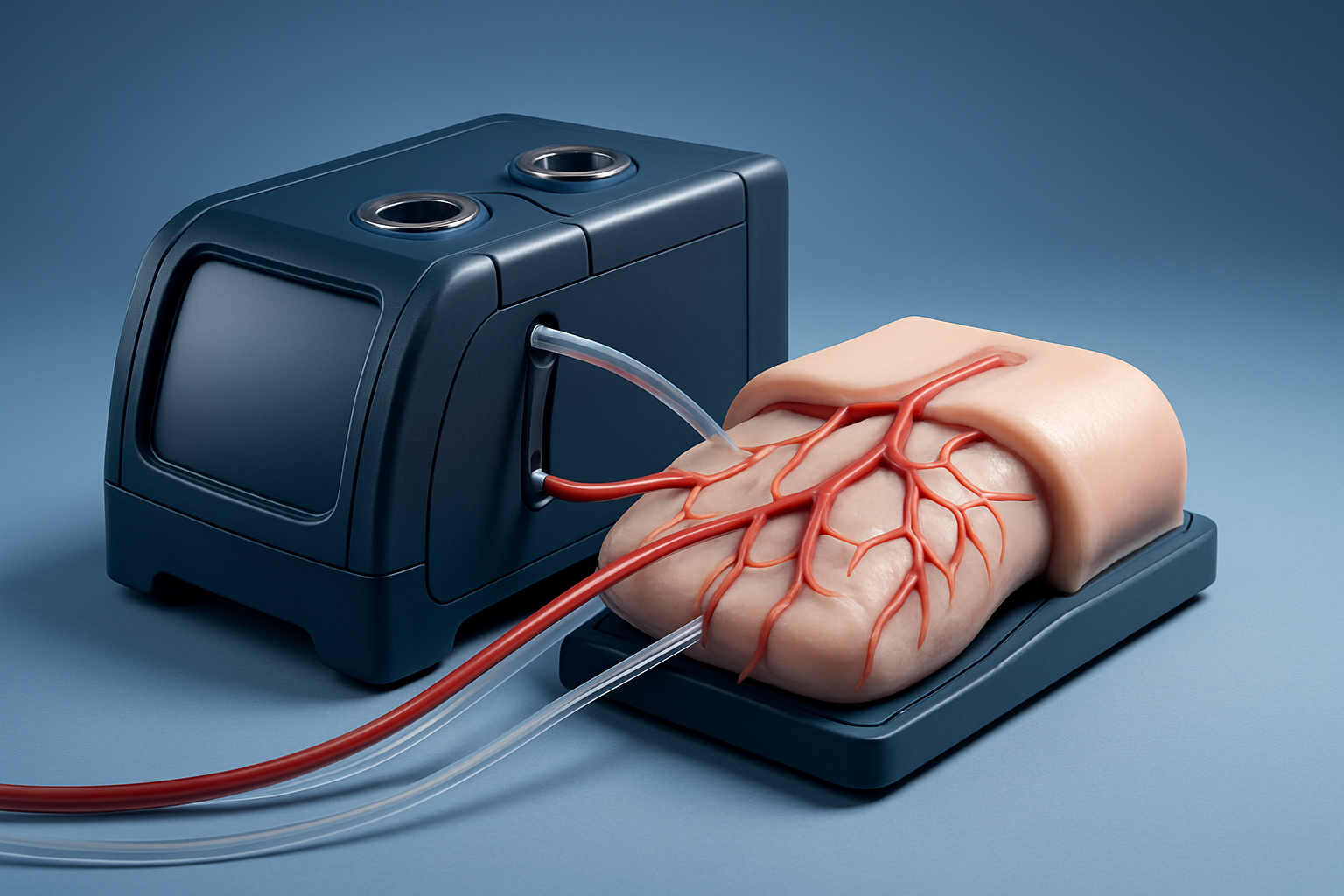
Final combined set visual
Complete ECMO training system
All‑in‑one bundle with AI program oversight.
Pricing at a glance
Modules are listed in EUR. The full ECMO set is listed in TL for local procurement.
Housing Product
Standalone enclosure for repeatable training rigs.
EUR 499
Vessel Integration Product
Lifelike vascular circuit with flow dynamics.
EUR 499
Final Combined Set
Best valueFull system with AI program oversight.
TL 9,990
Entire Automation Service
AI automation, validation alignment, production readiness.
Scope‑based
Atlas MedTech Services · Service Lane
Monthly consultancy
EUR 499 / monthGovernance, validation readiness, and rollout support.
End‑to‑end production solutions
Automation setup, audit readiness, and consultancy services.
Need a custom build?
Align pricing, service lines, and portfolio outcomes with our ECMO delivery team.
We move from the medtech-highlight pipeline to services-overview execution with audit-ready systems, measurable milestones, and clear stakeholder alignment.
Speed
Rapid pilots
Scope
Six verticals
Compliance
Audit-ready
Palette guidance
Use primary for decision CTA focus, secondary for service lanes, and moderate for compliance signals to keep the system consistent.
Impact pillars
System build
Modular systems designed for growth, validation, and dependable uptime.
Coverage
Medtech, automation, agriculture, and digital operations aligned.
Compliance
Validation, documentation, and QA aligned with regulated environments.
Need a tailored impact roadmap?
Book a discovery session for a focused technical plan.
Atlas Global Technologies builds validated, automation-ready systems that move from concept to audit-ready delivery across healthcare and industry.
A simple, repeatable path from mission to measurable outcomes.
Define the clinical, technical, and regulatory goals for success.
Engineer AI-enabled platforms, devices, and workflows with validation in mind.
Deploy across MedTech, industrial, and commercial partners for measurable ROI.
NEWS / JOURNAL
Direct updates on MedTech innovation, AI automation, and multidisciplinary engineering across Atlas Global Technologies.
Validation-ready workflows now integrate AI monitoring, real-time data capture, and hospital feedback for faster readiness.
Read updateA multi-stage automation stack delivered traceability and validation reporting aligned with medical production standards.
Read updateAtlas teams partnered with local growers to deploy sensor-driven monitoring and functional bio-farming outputs.
Read updateFAQ
Atlas Global Technologies supports complex B2B engagements across medtech, engineering, and AI automation. Here are the questions we hear most from executive teams and technical leads.
Need a tailored response?
Share your scope, timelines, compliance needs, and procurement pathway. We will respond with a clear consultation flow and next steps.
Contact Atlas Global TechnologiesMedtech, biomedical engineering, AI automation, industrial production, agriculture technology, digital brand management, and automotive tuning programs.
Yes. We deliver simulation systems, validation-ready documentation, and training support tailored to clinical education and operational goals.
It creates a realistic ECMO practice environment with tissue and artificial vessel integration for clinical training, assessment, and protocol readiness.
Yes. Modular housing, vessel configurations, and training pathways can be adapted to your protocols and regional compliance needs.
We provide validation guides, quality records, setup manuals, and audit-ready documentation aligned with regulated environments.
We begin with discovery, align requirements and compliance scope, then deliver a timeline, budget, and implementation plan.